Description
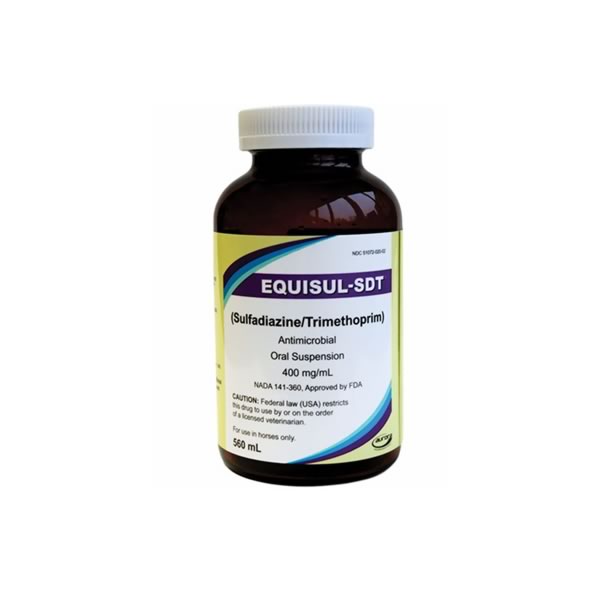
Equisul – SDT. This product is manufactured by Aurora Pharmaceutical.
Active Ingredients: Each mL of EQUISUL-SDT contains 400 mg combined active ingredients (333 mg sulfadiazine and 67 mg trimethoprim) in an aqueous suspension.
Directions for User: Shake well before use. Follow veterinarians instructions for use. The manufacturer recommends the following: Administer EQUISUL-SDT orally at the dosage of 24 mg combined active ingredients per kilogram body weight (10.9 mg/lb) twice daily for 10 days. EQUISUL-SDT can be administered by volume at 2.7 mL per 45.4 kg (2.7 mL/100 lb) body weight.
Special Precautions: Not for use in humans. For use in animals only. Keep this and all drugs out of the reach of children. Consult a physician in the case of accidental human exposure. Antimicrobial drugs, including sulfonamides, can cause mild to severe allergic reactions in some individuals. Avoid direct contact of the product with the skin, eyes, mouth, and clothing. Persons with a known sensitivity to sulfonamides or trimethoprim should avoid exposure to this product. If an allergic reaction occurs (e.g., skin rash, hives, difficulty breathing, facial swelling) seek medical attention.