Description
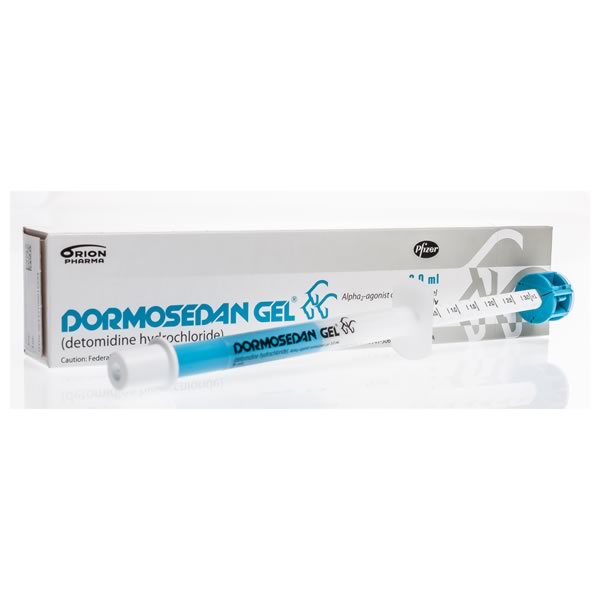
Medication should be delivered beneath the tongue (sublingually) and is not meant to be swallowed. For equine use only. Detomidine, the active ingredient in Dormosedan, is designated as a class 3 substance by the ARCI.
Active Ingredients: detomidine hydrochloride
Directions for User: Administer dose based on weight of horse. The dosing syringe delivers the medication in 0.25ml increments and is intended for one-time use and then should be discarded. The horse should be kept in a quiet, comfortable environment before, during and after treatment. It is important to also withhold food and water until the sedative effects have worn off.
Special Precautions: Intravenous potentiated sulfonamides should not be used in anesthetized or sedated horses, as potentially fatal dysrhythmias may occur. Do not use DORMOSEDAN GEL in horses with pre-existing atrioventricular (AV) or sinoatrial (SA) blocks, cardiovascular disease, respiratory disorders, liver or kidney diseases, or in conditions of shock, severe debilitation or stress due to extreme heat, cold, fatigue or high altitude. Appropriate precautions should be taken while handling and using gel-dosing syringes, as DORMOSEDAN GEL can be absorbed following direct exposure to skin, eyes or mouth, and may cause irritation. The use of impermeable gloves is advised.